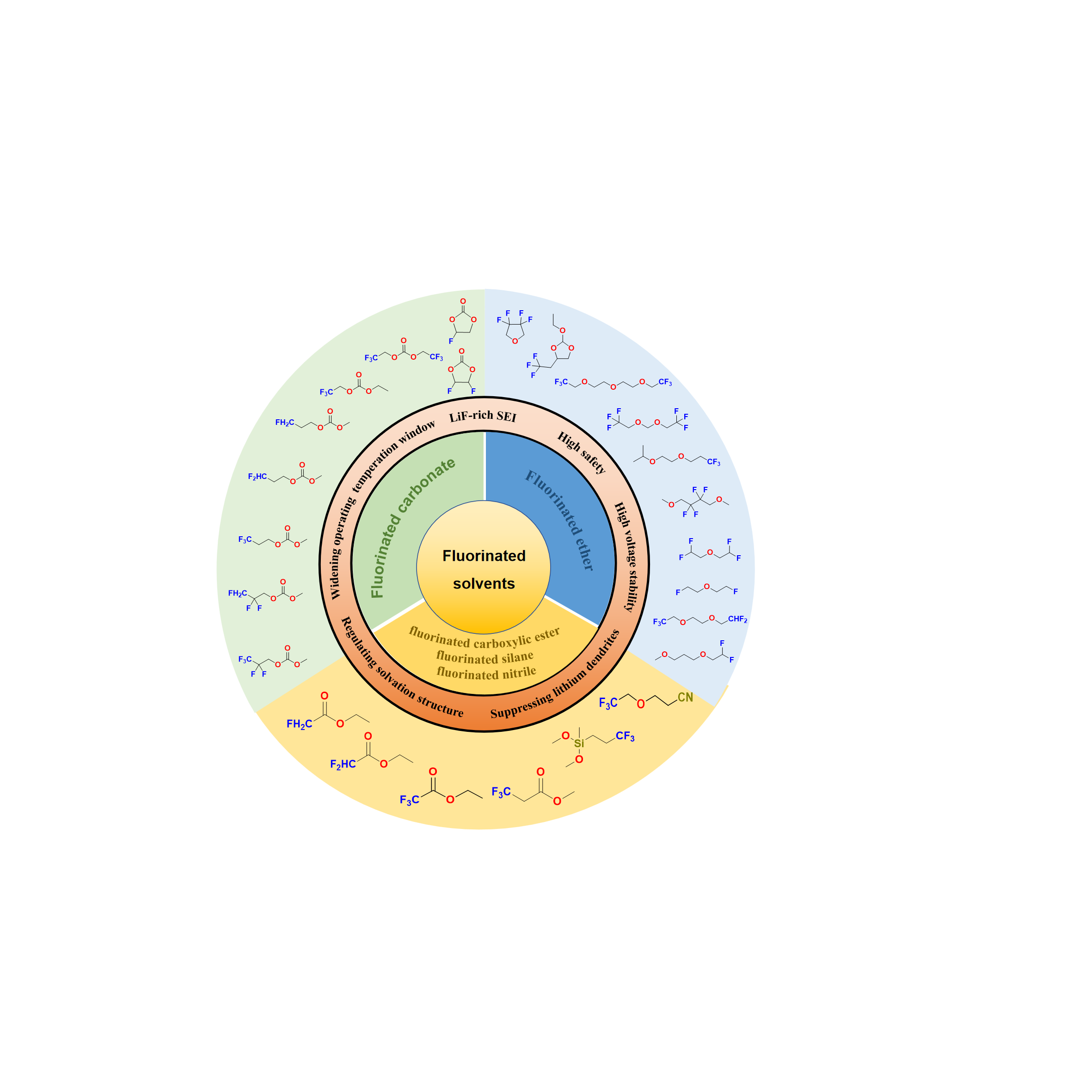
1 引言
2 氟代碳酸酯类溶剂
表1 氟代碳酸酯类电解液的电池性能Table 1 Cell performance of fluorinated carbonate based electrolytes |
| Fluorinated carbonate | Electrolyte | Batteries | Cycling Condition | Cell Performance | Ref |
|---|---|---|---|---|---|
| Fluorinated cyclic carbonate | 1.2 M LiPF6-FEC | Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 1050 h | 34 |
| 1 M LiPF6-FEC∶EMC(1∶3, by vol) | Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 900 h | 35 | |
| Li||NMC622 | 2.7~4.3 V, 1 C | 70%@250 cycles | |||
| 1 M LiPF6-FEC∶DMC(1∶4, by vol) | Li||Li | 2 mA·cm-2, 3.3 mAh·cm-2 | 3600 h | 36 | |
| 1 M LiPF6-DFEC∶DEC(1∶1, by vol) | Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 800 h | 38 | |
| Li||NMC811 | 2.8~4.5 V, 0.2 C | 91%@300 cycles | |||
| 7 M LiFSI-FEC | Li||Li | 0.25 mA·cm-2, 0.25 mAh·cm-2 | 300 h | 39 | |
| Li||LNMO | 3.0~5.0 V, 0.36 C | 78%@130 cycles | |||
| 2 M LiPF6-EC∶DME (1∶1, by vol)+50% FEC | Li||Cu | 0.2 mA·cm-2 | CE: 98%@1066 h | 40 | |
| 4 M LiTFSI+0.5 M LiDFOB-FEC∶DMC(3∶7, by vol) | Li||Cu | 0.5 mA·cm-2 | CE: 98%@900 cycles | 41 | |
| Li||LNMO | 3.5~4.9 V, 1 C | 88.5%@500 cycles | |||
| Li soaked in FEC, 1 M LiPF6-ACN | Li||Li | 0.1 mA·cm-2 | 1500 h | 42 | |
| Li||LiFePO4 | 2.2~4.2 V, 0.2 C | 82%@500 cycles | |||
| Li soaked in FEC, 1 M LiPF6-EC∶DEC(2∶1, by vol) | Li||NMC | 3.0~4.3 V, 0.5 C | 68.2%@120 cycles | 43 | |
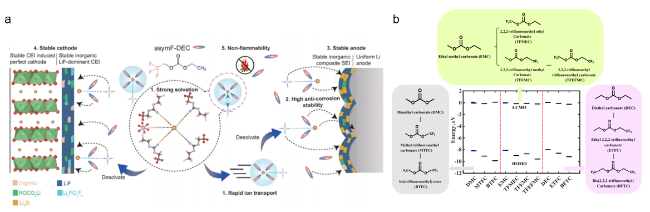
| Fluorinated linear carbonate | 1 M LiPF6+0.02 M LiDFOB- FEC∶FDEC∶TTE(2∶6∶2, by vol) | Li||LiCoMnO4 | 3.0~5.3 V, 1 C | 80%@1000 cycles | 44 |
| 1 M LiPF6-asymFDEC∶FEC∶VC (8∶2∶0.5, by vol) | Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 98.97% | 45 | |
| Li||NMC811 | 3.0~4.35 V, 0.5 C | 80%@240 cycles | |||
| 1 M LiPF6-FEC∶FEMC∶TTE (2∶6∶2, by vol) | Li||NMC811 | 2.7~4.3 V, 0.5 C | 90%@400 cycles | 46 |
2.1 氟代环状碳酸酯类溶剂
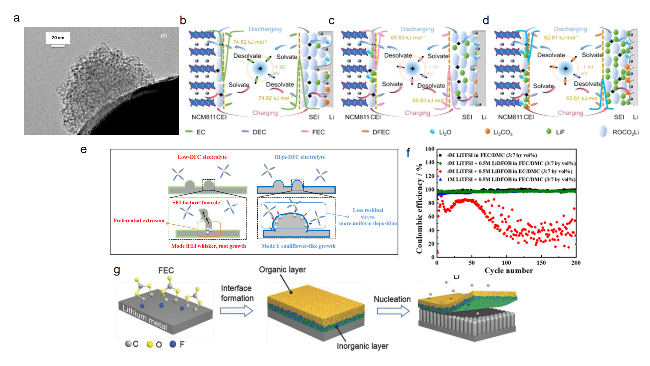
图1 (a) 1.2 mol/L LiPF6-FEC电解液的锂金属电极的高倍TEM图像[34]; (b) 1 mol/L LiPF6-EC∶DEC(1∶1,体积比)、(c) 1 mol/L LiPF6-FEC∶DEC(1∶1,体积比)、(d)1 mol/L LiPF6-DFEC/DEC(1∶1,体积比)电解液中锂离子溶剂化鞘动态演化示意图[38];(e) 高浓度 FEC 基电解液对 SEI 层主要贡献;(f) FEC 电解液及不含 FEC 电解液的长循环库仑效率[41];(g) 锂金属负极经 FEC 处理后形成双层膜的示意图[43]Fig. 1 (a) HR-TEM image of lithium metal anode in the electrolyte of 1.2 mol/L LiPF6-FEC[34]. Copyright 2023, American Chemical Society ; Schematic diagram of the dynamic evolution of Li+ solvated sheath in the electrolytes of (b) 1 M LiPF6-EC/DEC(1∶1, by vol), (c) 1 mol/L LiPF6-FEC/DEC(1∶1, by vol), (d) 1 mol/L LiPF6-DFEC/DEC(1∶1, by vol)[38], Copyright 2023, American Chemical Society; (e) The main contribution of FEC-based HCE to the SEI layer, Copyright 2021, Wiley-VCH GmbH; (f) Coulombic efficiency of FEC electrolytes and FEC-free electrolytes[41]. Copyright 2020, American Chemical Society; (g) The formation of a double-layer film on lithium metal anode after FEC treatment[43]. Copyright 2018, WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim |
2.2 氟代线性碳酸酯类溶剂
3 氟代醚类溶剂
表2 不同氟代醚类电解液的电池性能Table 2 Cell performance of fluorinated ether based electrolytes |
| Fluoroether solvents | Electrolyte | Batteries | Cycling Conditions | Cell Performance | Ref |
|---|---|---|---|---|---|
| Fluorinated cyclic ether | 1 M LiFSI-DME∶TFF (1∶2.7, by vol) | Li||Cu | 5 mA·cm-2, 0.5 mAh·cm-2 | CE: 99.4% | 51 |
| Li||NMC811 | 2.8~4.3 V, 0.2 CC/0.3 CD | 75%@300 cycles | |||
| 1 M LiDFOB+0.4 M LiBF4-DME∶HFTFP (1∶4, by vol) | Li||NMC811 | 2.8~4.3 V, 0.2 CC/0.3 CD | 80%@190 cycles | 53 | |
| 2 M LiFSI-cFTOF | Li||NMC811 | 3.0~4.3 V, 0.5 C | 100%@112 cycles | 52 | |
| 2 M LiFSI-DTDL | Li||NMC811 | 2.8~4.2 V, 0.5 C | 84%@200 cycles | 54 | |
| 1.5 M LiFSI-TTD∶DME (8∶2, by vol) | Li||NMC811 | 2.8~4.7 V, 0.5 C | 80%@100 cycles | 55 | |
| Fluorinated linear ether | 1 M LiFSI-E3F1 | Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 700 h | 56 |
| Li||Cu | 0.5 mA·cm-2 | CE: 98.9% | |||
| 2 M LiFSI-TTME∶DME (4∶1, by vol) | Li||Li | 0.5 mA·cm-2, 1 mAh·cm-2 | 3200 h | 57 | |
| Li||Cu | 1 mA·cm-2 | CE: 99.3% | |||
| Li||LCO | 3.0~4.5 V, 0.3 C | 85%@170 cycles | |||
| 2 M LiFSI-PXEO-CF3 | Li||Cu | 5 mA·cm-2 | CE: 99.2% | 58 | |
| Li||SPAN | 1.0~3.0 V, 4 C | 89.8%@1500 cycles | |||
| 2 M LiFSI-TMDMP | Li||Li | 1 mA·cm-2, 1 mA·cm-2 | 1600 h | 59 | |
| Li||Cu | 1 mA·cm-2 | CE: 99.6% | |||
| Li||NMC811 | 2.8~4.4 V, 0.1 C | 81%@200 cycles | |||
| 1 M LiFSI-FDMB | Li||NMC532 | 3.0~4.2 V, 0.3 C | 90%@420 cycles | 61 | |
| 1 M LiFSI-FDMH∶DME (6∶1, by vol) | Li||Cu | 1 mA·cm-2 | CE: 99.5% | 62 | |
| Li||NMC532 | 3.0~4.2 V, 0.3 C | 84%@250 cycles | |||
| Partial fluorinated linear ether | 1.4 M LiFSI-BDE∶DME (1∶6, by vol) | Li||Li | 0.5 mA·cm-2, 1 mAh·cm-2 | 2000 h | 65 |
| Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 99.6% | |||
| 1 M LiFSI-BFE | Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 99.8% | 66 | |
| LiFSI∶FDEE∶TTE (1∶1.6∶3, by mol) | Li||Li | 10 mA·cm-2, 1 mAh·cm-2 | 800 h | 67 | |
| Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 99.4% | |||
| Li||NMC811 | 2.8~4.7 V, 0.3 C | 92%@150 cycles | |||
| 1.2 M LiFSI-F4DEE | Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 99.5% | 68 | |
| 1.2 M LiFSI-F5DEE | Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 99.9% | 68 | |
| Li||NMC811 | 2.8~4.4 V, 0.1 CC/0.3 CD | 80%@270 cycles | |||
| 2.1 M LiFSI-F2EMP | Li||Cu | 0.5 mA·cm-2, 1 mAh·cm-2 | CE: 99.35% | 69 | |
| Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 1200 h |
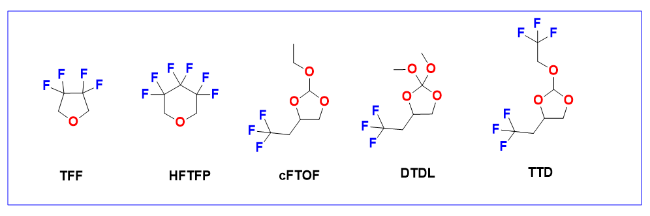
3.1 氟代环状醚类溶剂
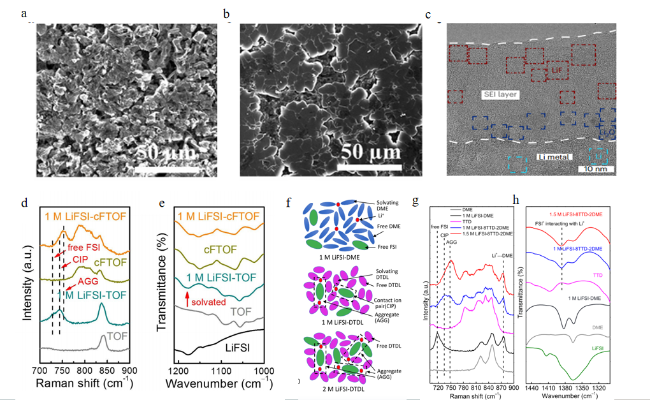
图5 (a) 1 mol/L LiFSI-DME循环30次后的沉积锂形态[51];(b) 1mol/L LiFSI-DME/TFF循环30次后的沉积锂形态[51];(c) D-HFTHP 中锂金属上的SEI的 Cryo-TEM 图像[53]; (d) 1 mol/L LiFSI-cFTOF电解液的拉曼光谱[52];(e) 1 mol/L LiFSI-cFTOF电解液的红外光谱[52] (f) DTDL电解液中锂离子配位结构示意图[54];(g) 1.5 mol/L LiFSI-TTD∶DME (8∶2, 体积比)电解液的拉曼光谱[55];(h) 1.5 mol/L LiFSI-TTD∶DME (8∶2, 体积比)电解液的红外光谱[55]Fig.5 Deposited Li morphology after 30 cycles at 0.75 mA cm-2 and 1.5 mAh cm-2 using different electrolytes[51] (a) 1 mol/L LiFSI-DME ; (b) 1 mol/L LiFSI-DME/TFF. Copyright 2023 , The Authors. Angewandte chemie international Edinon published by Wiley-VCH GmbH ; (c) Cryo-TEM image of SEI on lithium metal in D-HFTHP[53]. Copyright 2024, The Author(s), under exclusive licence to Springer Nature Limited; (d) Raman spectra of 1mol/L LiFSI-cFTOF electrolyte[52]; (e) FT-IR spectra of 1mol/L LiFSI-cFTOF electrolyte[52]. Copyright 2022 , The Authors. Angewandte chemie linternational Edition published by Wiley-VCH GmbH (f) Schematic diagrams of lithium-ion coordination structure in DTDL electrolyte[54]. Copyright 2022, The Author(s); (g) Raman spectra of 1.5 mol/L LiFSI-TTD∶DME (8∶2, by vol) electrolytes[55]; (h) FT-IR spectra of 1.5 mol/L LiFSI-TTD∶DME (8∶2, by vol) electrolyte[55]. Copyright 2022, American Chemical Society |
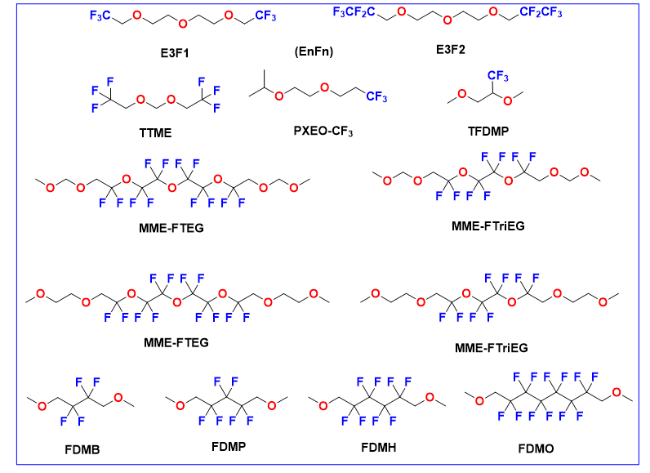
3.2 氟代线性醚类溶剂
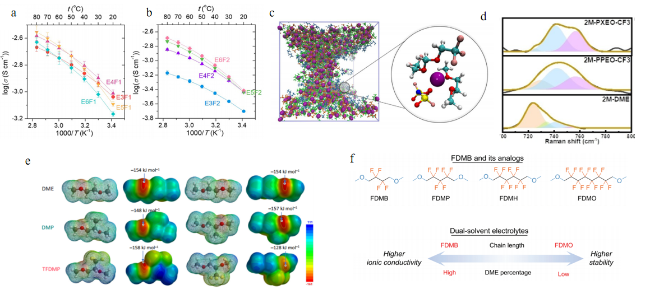
图7 (a) EnF1系列化合物离子电导率数据图[56];(b) EnF2系列化合物离子电导率数据图[56];(c)TTME电解液的溶剂化结构[57];(d) PXEO-CF3电解液的拉曼光谱[58];(e) TFDMP与DME和DMP的静电势能对比图[59];(f) FDMH分子设计原理[62]Fig.7 (a) Ionic conductivity of EnF1 based electrolytes[56] ; (b) Ionic conductivity of EnF2 based electrolytes[56]. Copyright 2021, American Chemical Society; (c) Solvation structure of TTME electrolyte[57]. Copyright 2023, Science Press and Dalian institute ofchemical Physics, Chinese Academy of Sciences. Published by ELSEViER B.V. and Science Pres. All rights reserved; (d) Raman spectra of PXEO-CF3 electrolyte[58]. Copyright 2024, Wiley-VCH GmbH; (e) Electrostatic potential (ESP) maps of DME, DMP and TFDMP solvents with front and back views,[59]. Copyright 2023, The Author(s); (f) Diagram of the design strategy for dual-solvent electrolytes using DME as the co-solvent[62]. Copyright 2021, Wiley-VCH GmbH |
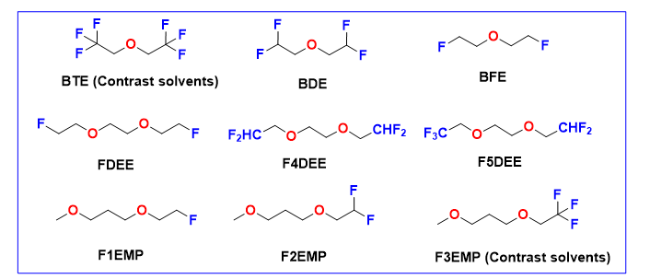
3.3 部分氟代醚类溶剂
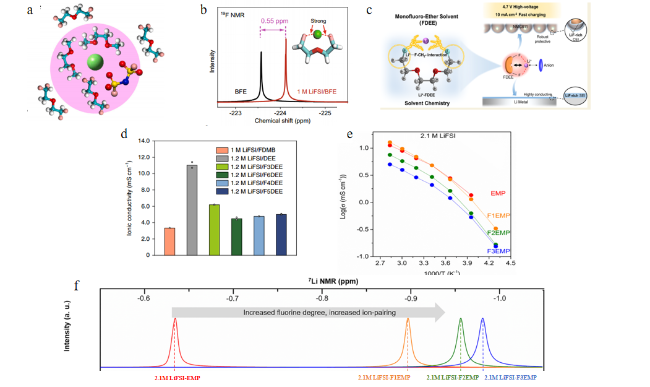
图9 (a) BDE/DME电解液溶剂化结构示意图[65];(b) BFE 基电解液19F NMR图谱[66];(c) FDEE溶剂化结构示意图[67];(d) 氟化-1,2-二乙氧基乙烷系列电解液离子电导率[68];(e) EMP和氟化EMP电解液离子电导率[69];(f) EMP和氟化EMP电解液的 7Li 核磁共振图[69]Fig.9 (a) The proposed unique solvation structure of BDE/DME electrolyte[65]. Copyright 2022, Elsevier Ltd. All rights reserved; (b) 19F NMR of BFE before and after the salt dissolution[66]. Copyright 2023, The Author(s); (c) Solvation structure of FDEE electrolyte[67]. Copyright 2023, American Chemical Society; (d) Ionic conductivity of fluorinated 1,2-diethoxyethane based electrolytes[68]. Copyright 2022, The Author(s); (e) Ionic conductivity of EMP and fluorinated EMP electrolytes[69]; (f) 7Li NMR of EMP and fluorinated EMP electrolytes[69]. Copyright 2024, American Chemical Society |
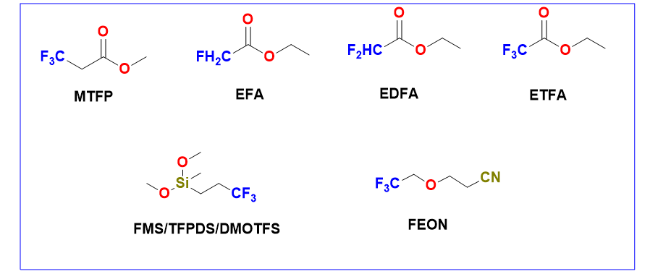
4 其他氟代溶剂
图10 氟代羧酸酯、氟代硅氧烷、氟代腈类分子的结构示意图Fig.10 Chemical structures of fluorinated carboxylic ester, fluorinated silane and fluorinated nitrile molecules |
表3 氟代羧酸酯、氟代硅烷、氟代腈类电解液的电池性能Table 3 Cell performance of fluorinated carboxylic ester, fluorinated silane, and fluorinated nitrile |
| Other fluorinated solvents | Electrolyte and Amount | Batteries | Cycling Condition | Cell Performance | Ref |
|---|---|---|---|---|---|
| Fluorinated carboxylic ester | 1 M LiPF6-MTFP∶FEC (9∶1, by vol) | Li||NMC811 | 4.5 V, 0.5 C | 80%@250 cycles | 71 |
| Fluorinated silane | 2.2 M LiFSI-DMOTFS | Li||NMC811 | 3~4.7 V, 0.5 C | 82.8%@180 cycles | 74 |
| 3 M LiFSI-FMS | Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 1200 h | 75 | |
| Li||Cu | 1 mA·cm-2, 1 mAh·cm-2 | CE: 99.1% | |||
| 1.5 M LiFSI-TFPDS | Li||LCO | 3~4.6 V, 0.5 C | 90%@320 cycles | 76 | |
| Fluorinated nitrile | 0.8 M LiTFSI+0.2 M LiDFOB-FEON∶FEC (1∶3, by vol) | Li||Li | 1 mA·cm-2, 1 mAh·cm-2 | 600 h | 79 |
| Li||Cu | 1 mA·cm-2, 0.5 mAh·cm-2 | CE: 98.6% |
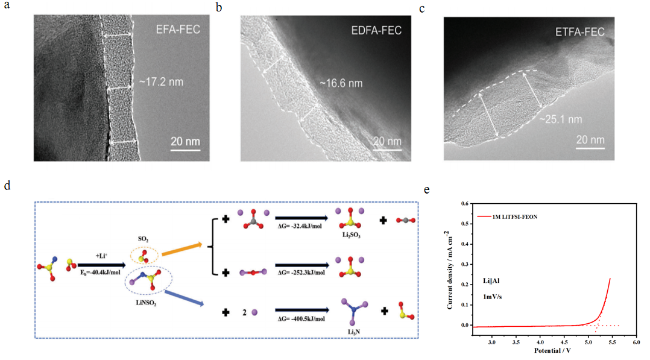
图11 TEM测试三种氟代羧酸酯SEI层形貌[72](a) EFA-FEC基电解液、(b) EDFA-FEC基电解液、(c) ETFA-FEC基电解液;(d) FSI--2F-的可能还原分解产物以及反应能(kJ·mol-1)[74];(e) FEON基电解液的LSV[78]Fig.11 TEM images of Gr electrode cycled in (a) EFA-FEC, (b) EDFA-FEC, and (c) ETFA-FEC[72]. Copyright 2023, Wiley-VCH GmbH; (d) Possible reductive decomposition products of FSI--2F- of together with the reaction energy (kJ·mol-1)[74]. Copyright 2022, The Author(s); (e)The LSV test of FEON-based electrolyte[78]. Copyright 2022, Royal Society of Chemistry |





 汪靖伦 研究员,博士生导师,主要从事电化学储能材料与器件、高性能锂离子电池有机和高分子功能材料的设计与合成等方向的研究,已在Adv. Funct. Mater., J. Energy Chem., ACS Sustainable Chem. Eng., J. Power Sources, Sci. China Chem., 《物理化学学报》《化工学报》等国内外刊物发表SCI论文50余篇,申请发明专利10余件。
汪靖伦 研究员,博士生导师,主要从事电化学储能材料与器件、高性能锂离子电池有机和高分子功能材料的设计与合成等方向的研究,已在Adv. Funct. Mater., J. Energy Chem., ACS Sustainable Chem. Eng., J. Power Sources, Sci. China Chem., 《物理化学学报》《化工学报》等国内外刊物发表SCI论文50余篇,申请发明专利10余件。