 PDF(11922 KB)
PDF(11922 KB)


 PDF(11922 KB)
PDF(11922 KB)
 PDF(11922 KB)
PDF(11922 KB)
酰胺电子等排体在先导化合物优化中的应用
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Application of Amide Bioisosteres in the Optimization of Lead Compounds
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}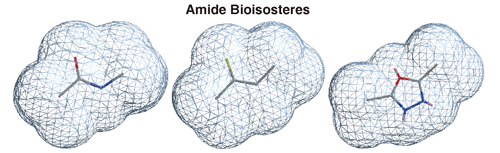
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |