 PDF(4291 KB)
PDF(4291 KB)


 PDF(4291 KB)
PDF(4291 KB)
 PDF(4291 KB)
PDF(4291 KB)
双网络水凝胶制备及其力学改性
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Preparation of Double Network Hydrogels and their Mechanical Modification
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}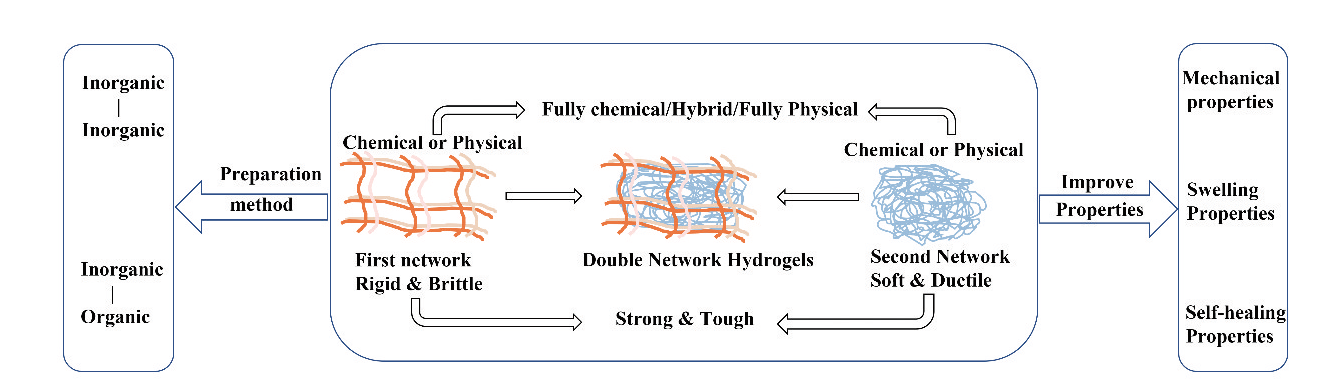
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |