 PDF(45467 KB)
PDF(45467 KB)


 PDF(45467 KB)
PDF(45467 KB)
 PDF(45467 KB)
PDF(45467 KB)
基于多齿钯化合物的磁性纳米颗粒催化材料的设计合成及应用
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Design, Synthesis and Application of Magnetic Nanoparticle Catalytic Materials Based on Multientate Palladium Compounds
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}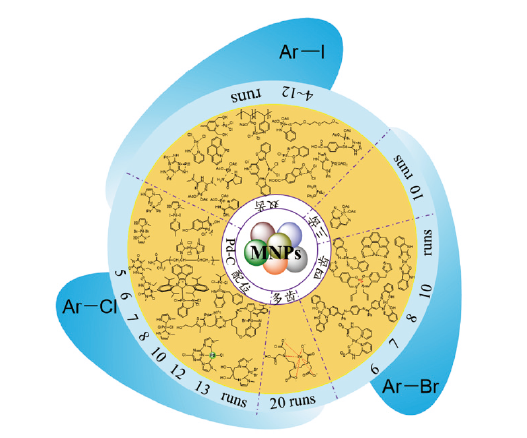
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |