 PDF(10158 KB)
PDF(10158 KB)


 PDF(10158 KB)
PDF(10158 KB)
 PDF(10158 KB)
PDF(10158 KB)
负载型廉价金属催化剂在低温催化氧化甲醛中的应用
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Application of Supported Non-Noble Metal Catalysts for Formaldehyde Oxidation at Low Temperature
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}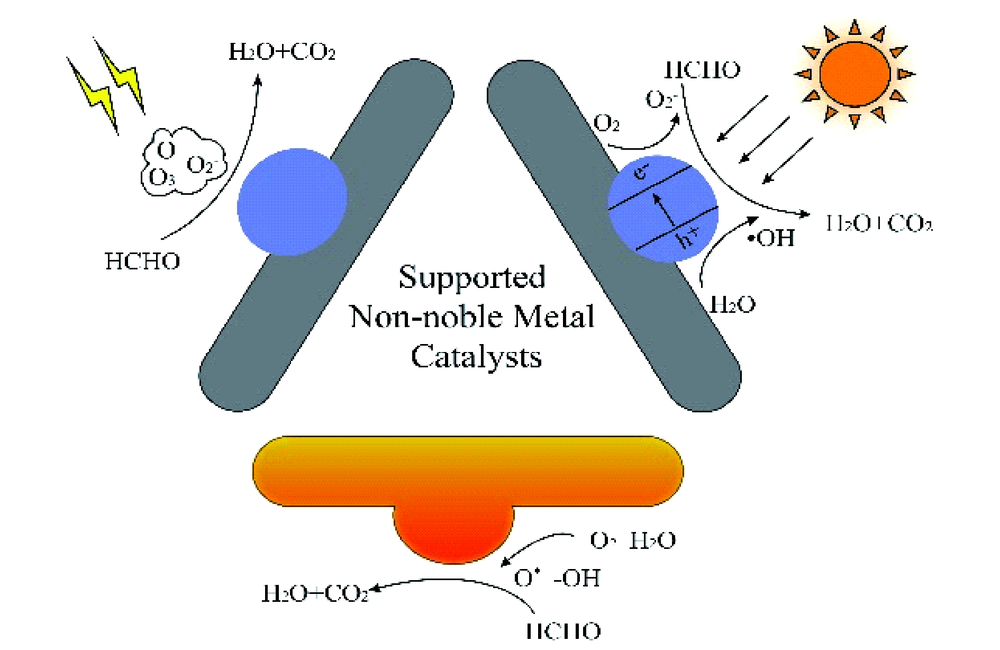
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |