 PDF(5636 KB)
PDF(5636 KB)


 PDF(5636 KB)
PDF(5636 KB)
 PDF(5636 KB)
PDF(5636 KB)
光电催化海水分解制氢
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Hydrogen Production by Photoelectrocatalytic Seawater Splitting
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}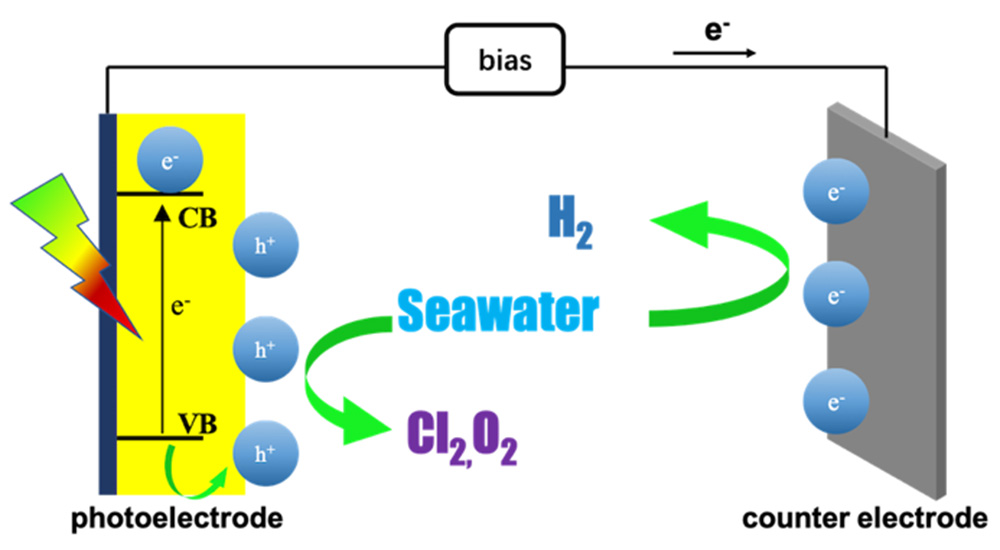
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |