 PDF(11299 KB)
PDF(11299 KB)


 PDF(11299 KB)
PDF(11299 KB)
 PDF(11299 KB)
PDF(11299 KB)
有机光致变色材料在快速可视化检测中的应用
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Applications of Organic Photochromic Materials in Rapid Visual Detection
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}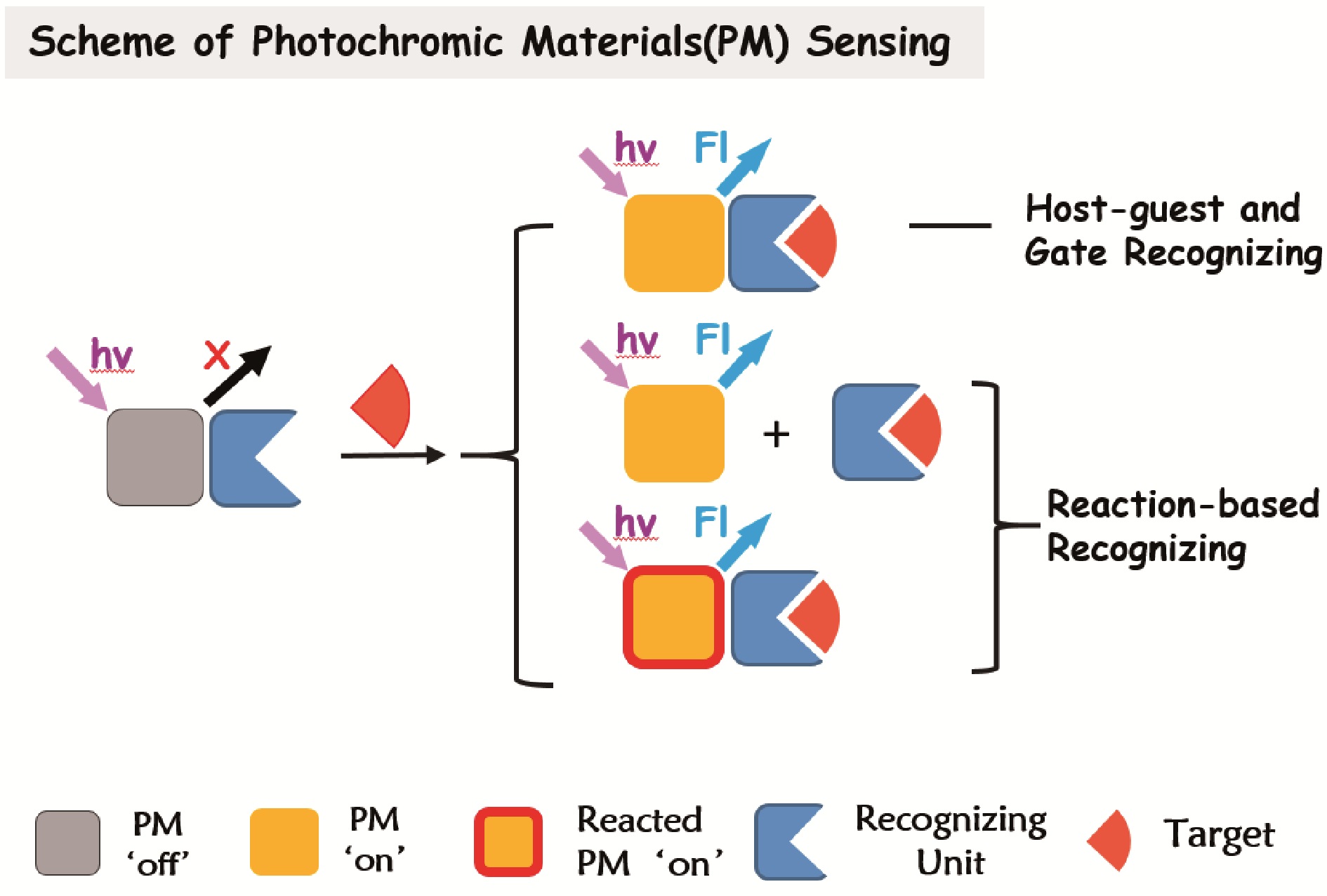
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |