 PDF(1172 KB)
PDF(1172 KB)


 PDF(1172 KB)
PDF(1172 KB)
 PDF(1172 KB)
PDF(1172 KB)
腈水解酶在医药中间体生物催化研究中的最新进展
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Recent Progress in the Application of Nitrilase in the Biocatalytic Synthesis of Pharmaceutical Intermediates
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}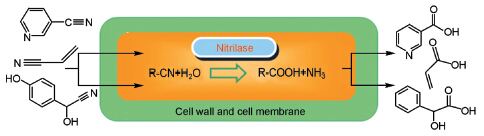
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |